Action required: Please refresh your browser
We have recently implemented some changes that require a hard refresh of your browser: Please hold down the CTRL-key and press the F5 key.
After a successful hard refresh, this message should not appear anymore.
More details about this topic are available here »
| Public advisory - Recall of one lot of JAMP Guanfacine XR 4 mg tablets due to contamination with foreign matter | ||||||||||
| By: PR Newswire Association LLC. - 12 Apr 2024 | Back to overview list |
|||||||||
|
OTTAWA, ON, April 12, 2024 /CNW/ - Summary
Images Affected products
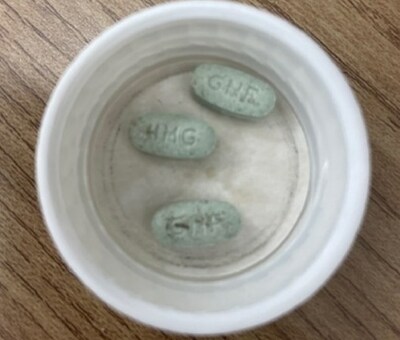
Issue Jamp Pharma Corporation is recalling one lot of JAMP Guanfacine extended release (XR) 4 mg tablets because some tablets may have been contaminated with foreign matter during manufacturing. The foreign matter causes brown or amber-coloured staining on the tablet. The foreign matter is composed of a combination of cellulose, lubricant oil, calcium, and/or iron oxide. These substances are not expected to pose any serious health risks; however, it is recommended that the medication be replaced if stains are identified. JAMP Guanfacine XR is a prescription drug used to treat Attention Deficit Hyperactivity Disorder (ADHD) in children and adolescents 6 to 17 years of age. The 4 mg tablet is green, oval shaped, with "GNF" stamped on one side and "4MG" stamped on the other. Health Canada is monitoring the company's recall and its implementation of any necessary corrective and preventative actions to stop this issue from reoccurring. The Department will inform the public if any new health risks are identified. What you should do
Additional information for health professionals:
Également disponible en français SOURCE Health Canada (HC) 
|
||||||||||
|
|
||||||||||
 | Back to overview list | |||||||||